A preconfigured, rapid deployment COVID-19 LIMS to manage COVID-19 diagnostic tests
Our team has further developed and preconfigured CloudLIMS to meet the urgent requirements of testing and research of COVID-19 in these unprecedented and challenging times,” said Arun Apte, Chief Executive Officer at CloudLIMS.
WILMINGTON, Del. (PRWEB)
May 02, 2020
CloudLIMS, a leading laboratory informatics provider, announces the release of a rapid deployment LIMS, preconfigured for COVID-19 testing. The LIMS can be quickly set up for COVID-19 testing, in 2 to 4 days, comes pre-loaded with FDA and CDC approved tests, report templates, and workflows to maximize testing and reporting capability of COVID-19 laboratories. This functionality enables COVID-19 diagnostic testing laboratories to manage a massive influx of COVID-19 specimens and to quickly turnaround test specimens and report results as per FDA and CDC guidelines.
Combined with the previous announcement of offering its flagship product, CloudLIMS Enterprise, free of charge to COVID-19 research and testing laboratories across the globe, CloudLIMS now extends support to such laboratories by offering its rapid deployment, preconfigured product. Read the previous press release here.
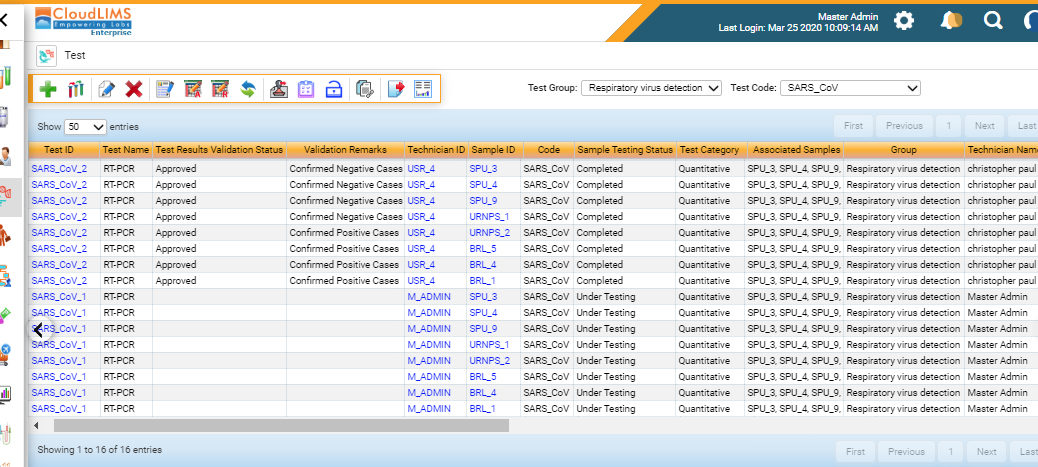
The demand for COVID-19 testing is growing exponentially with over 3 million confirmed cases globally. CloudLIMS, an in the cloud COVID-19 LIMS, is preconfigured with COVID-19 specific tests, such as RT-PCR, serological tests, workflows, and reporting templates, enabling laboratories to go-live in less than a week. CloudLIMS can be seamlessly configured to support new and upcoming test methods, assays, and reporting requirements, for COVID-19 and other infectious diseases, avoiding costly and time-consuming customization. CloudLIMS’ client portal enables physicians to place SARS-CoV-2 diagnostic test requests, track request status in real-time, download test reports, minimizing the turnaround time.
CloudLIMS’ REST API is preconfigured to support integration with COVID-19 testing instruments, such as RT-PCR, NGS instruments. Furthermore, the Rest API supports integration with third-party software such as EMR, EHR, LIS, patient registries, and clinical billing software. CloudLIMS seamlessly integrates with the reporting system of CDC/CSTE/FEMA and other e-reporting tools to automate the electronic reporting of results.
CloudLIMS enables laboratories to securely manage COVID-19 specimens, anonymize patient data, tests and test results, ensure quality control, schedule instrument calibration, manage training and competency of laboratory staff handling biohazardous specimens. CloudLIMS’ COVID-19 LIMS also enables clinical laboratories to follow regulatory guidelines and standards such as CLIA, HIPAA, ISO 15189:2012.
CloudLIMS can be deployed in less than a week and has the potential to maximize the testing and reporting capability of nascent and existing COVID-19 testing laboratories while maintaining FDA, CDC, and other regulatory guidelines. We hope to empower a greater number of laboratories to accelerate COVID-19 testing and research and help the world overcome the global health crisis,” he continued.
About CloudLIMS
Established in 2013, CloudLIMS is an energetic team of professionals producing cutting edge laboratory software solutions, such as sample management software and LIMS. For more information, please visit: http://www.cloudlims.com.
Share article on social media or email: